metadata
Title
Insect species recorded in sugarcane fields of Khon Kaen Province, Thailand, over three seasons in 2012
Running title
Insect records for sugarcane fields
Authors
Itsarapong Voraphab1, Yupa Hanboonsong1, Youichi Kobori2, Hiroaki Ikeda3 and Takeshi Osawa34*
1: Division of Entomology,
Faculty of Agriculture, Khon Kaen University,
Khon Kaen 40002, Thailand
2: Japan International Research Center for Agricultural Sciences, Japan
3: National Institute for Agro-Environmental Sciences, Japan
4: Japan node of the Global Biodiversity Information Facility
*: Corresponding author:
FAX: +81.78.803.8199
E-mail: arosawa@gmail.com
Abstract
The diversity of insect species in cultivated fields provides a number of ecosystem benefits, including natural pest control and pollination. However, current knowledge of the insect fauna in agricultural landscapes is extremely limited when compared with information available for other ecosystems, such as forests and rivers. Here, we present data on insect diversity, including functional feeding group diversity, in sugarcane fields of Khon Kaen Province, Thailand. Sugarcane is a major Thai crop. We collected insects using light trap systems located in 16 sugarcane field plots. Each light trap was deployed in an individual 0.16 ha plot. Trapping was conducted over three seasons, including both dry and rainy periods, in March, June, and October of 2012. On each trapping day, we collected insects in the evening from 18:00 to 19:30. The traps yielded a total of 98,423 individuals including 143 species belonging to 26 families. We classified collections into five functional feeding groups: herbivores, predators, decomposers, parasitoids, and pollinators. The dominant functional species were herbivores, followed in rank order by predators; most individuals in the traps were predatory species. We assembled an insect fauna data set that will be useful in the study of southeast Asian agricultural ecosystems and provided the data in the Darwin Core Archive format.
Keywords
- agricultural ecosystem
- decomposer
- herbivore
- parasitoid
- pollinator
- predator
Metadata
Introduction
Humans had ca. 1.5 × 109 ha of land area under cultivation globally in 2008 (FAO 2011) so that agricultural landscapes occupied ca. 11% of all terrestrial surfaces. Biodiversity in these agricultural landscapes contributes significantly to total terrestrial species richness, and plays a major role in the provisioning of ecosystem services (Bianchi et al. 2006; Frison et al. 2011). The diversity of insect species is especially relevant to the provisioning of a number of food production-related benefits provided by agricultural ecosystems, such as natural pest control (Landis et al. 2000; Thrupp 2000; Bianchi et al. 2006) and pollination (Vergara and Badano 2009; Garibaldi et al. 2013). However, relatively sparse collections of biodiversity data are currently available for agricultural ecosystems when compared with information available for other ecosystems, such as forests and rivers. Furthermore, records for common species (i.e., those that are not endangered or rare) on cultivated lands are also severely limited (Yamamoto and Kusumoto 2008; Osawa et al. 2013). Basic biodiversity records are essential for the compilation of baseline databases and for deepening our understanding of agricultural ecosystems, especially in diversity hotspots, which include the arable lands of southeast (SE) Asia. Biodiversity data for agricultural landscapes in SE Asia are particularly scarce, partly because of the logistical difficulties of data collection, but also because arable lands do not have protected status within national or provincial parks, where data gathering efforts are typically concentrated.
As indicated above, insects provide beneficial services in agricultural systems. Some have negative effects, such as grazing on crops and acting as vectors of diseases (Purcell & Almeida 2005). Insect diversity data will contribute to research aimed towards improving agricultural yield (e.g. Vergara and Badano 2009). Here, we present insect species records (including information on functional feeding categories) for sugarcane fields in Khon Kaen Province, Thailand. Thailand contributes significantly to agricultural production in SE Asia.
The data provided include information on insect functional feeding groups that are themselves related to crop yield efficiency: herbivores (pests), pollinators, predators, parasitoids, and decomposers. Previous studies showed that functional diversity itself is strongly related to several ecosystem functions and services (Zac et al. 1994; Tilman et al. 1997; Sasaki et al. 2014); thus, records of insect functional diversity might also contribute to the study of topics other than agricultural yield efficiency. Therefore, our data set may be helpful to researchers with diverse interests in terrestrial landscapes. We provide information sorted by season, making the data more relevant to users of our data set. Our data collection procedures allow cross comparisons with investigations conducted in other regions worldwide, especially in sugarcane growing areas. Sugarcane is major crop across tropical regions of the planet, including Brazil and India (FAO: http://faostat.fao.org; accessed 29/12/2014). Our comprehensive data set may be usefully extrapolated to parts of the world with data deficiencies.
Study area
Sugarcane fields of Khon Kaen Province, located in northeastern Thailand provided the study area (Fig. 1). In 2012, Thailand was the fourth largest producer and second largest exporter of sugarcane worldwide (FAO: http://faostat.fao.org; accessed 29/11/2014).
Site information
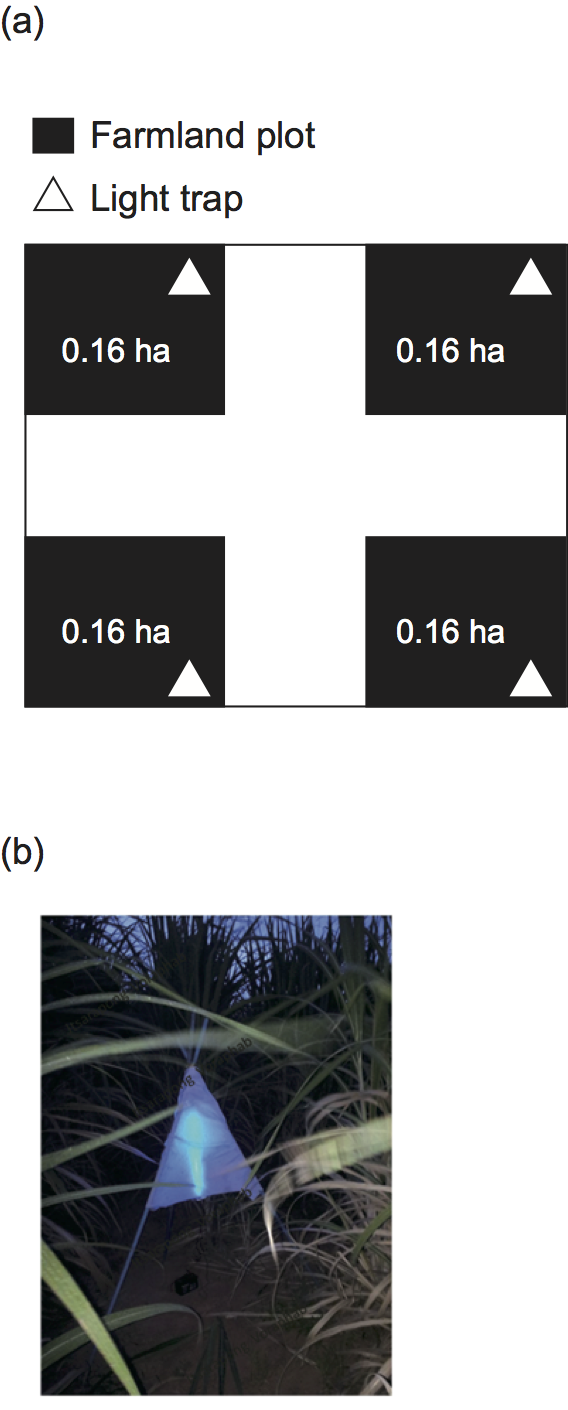
We focused on the Khon Kaen III sugarcane variety in four Muang District fields in an area enclosed within the following four sets of coordinates: 16° 18.85′ N, 102° 49.644′ E; 16° 18.698′ N, 102° 49.303′ E; 16° 13.989′ N, 102° 48.56′ E; 16° 13.5′ N, 102° 48.335′ E (WGS84 coordinate system). Four replicates were established at each site, giving a total 16 research plots across the district. Each plot covered 0.16 ha (one Thai rai unit; Fig. 2).
Sampling methods
We collected insect species within the plots with black light-blue light traps (20 W light bulbs) covered with 100 × 100 cm of thin white cloth (Fig. 2). Using a net, we captured all of the insects attracted to the lights. The sampling effort required of this procedure was quite tractable. Sugarcane grows rapidly and is able to develop from seedling to mature phases in a few months, reaching a final height of > 3 m. Sweeping and beating to collect insects in the cane fields through the seasons required unacceptably high levels of effort. The light trap sampling system is very straightforward compared with direct collection procedures, such as sweeping and beating. The light traps were located 2 m distant from the plot edges, and 1 m above ground level. The lights shone from 18:00 through 19:30 on each sampling day. Insect collection was only performed under very uniform conditions by avoiding days with rain or wind; the temperature in this part of Thailand varies very little throughout the year.
Only rainfall irrigates sugarcane in this area with cane planted in the dry season (October–December), and harvested the following year in the November–February. We made one insect collection in each plot during each of three phases of sugarcane growth: tillering (March, 3 months old), stalk elongation (June, 6 months old), and harvesting (October, 10 months old). Thus, our collections spanned the typical regional cycle of the sugarcane planting, and included seasonal changes that are likely to have influenced the composition of insect communities. All of the insects were counted and identified in the laboratory.
Contributors
A. Dataset owner and contact person
Name: Yupa Hanboonsong
Affiliation:
Division of Entomology, Faculty of Agriculture, Khon Kaen University
Address: Khon Kaen 40002, Thailand
Mail: yupa_han@kku.ac.th
B. Data management and release on GBIF network
Name: Takeshi OSAWA
Affiliation:
National Institute for Agro-Environmental Sciences / Japan node of the Global
Biodiversity Information Facility (JBIF)
Taxonomy and systematics
All species were identified by the authors and divided into respective functional feeding groups based on Triplehorn et al. (2005) and by comparison with specimens of the insect museum at Khon Kaen University. If we could not obtain sufficient reference information for proper identification, we determined the function of each specimen using relevant morphotypes such as the snout.
Taxonomic ranks
| Class | Order | Family | |
|---|---|---|---|
| Herbivores | Insecta | Hemiptera | Cicadellidae |
| Delphacidae | |||
| Pentatomidae | |||
| Coleoptera | Scarabaeidae | ||
| Tenebrionidae | |||
| Elateridae | |||
| Cuculidae | |||
| Chrysomelidae | |||
| Lepidoptera | Pyralidae | ||
| Erebidae | |||
| Noctuidae | |||
| Predators | Insecta | Hemiptera | Nabidae |
| Miridae | |||
| Reduviidae | |||
| Coleoptera | Staphylinidae | ||
| Carabidae | |||
| Coccinellidae | |||
| Orthoptera | Mantidae | ||
| Odonata | Libellulidae | ||
| Lestidae | |||
| Neuroptera | Chrysopidae | ||
| Dermaptera | Labiidae | ||
| Parasitoids | Insecta | Hymenoptera | Braconidae |
| Diptera | Tachinidae | ||
| Decomposers | Insecta | Dictyoptera | Blattellidae |
| Blaberidae | |||
| Pollinators | Insecta | Lepidoptera | Pyralidae |
Data format
All datasets were formatted using the procedures provided by the Darwin Core Archives, which are defined by the Biodiversity Information Standards (BIS) or Taxonomic Databases Working Group (TDWG: http://www.tdwg.org/). The protocols provide data standards for publishing and integrating biodiversity information (Vieglais 2012; Wieczorek et al. 2012). The Darwin Core, which allows users to record the occurrence of various organisms worldwide (e.g., specimen records or observational records with associated environmental information), is the de facto standard for describing species occurrences (Osawa et al. 2011), and is now used in the Ecological Research Data Paper Archives (Osawa, 2013). The aim of this initiative was to maintain a standard that is as simple and open as possible, and to develop terms only in cases of shared demand (Wieczorek et al. 2012). The Darwin Core has a relatively long history of community development, and is widely used in the field of biodiversity informatics (Canhos et al. 2004). When we were unable to identify a specimen to species name, we entered 'sp' or 'sp #' in the records.
Datasets information
Object name: Insect community records for
sugarcane fields over three seasons in Khon Kaen Province, Thailand, 2012
Character encoding: UTF-8
Format name: Darwin Core Archive format
Publication date of data: 2014/5/2
Language: English
Acknowledgements
We would like to thank Dr. D. Sprague for proofreading English. The Cluster Holistic Watershed Management of the National Research University Program, Khon Kaen University, Thailand supported this research; the Environment Research and Technology Development Fund (S9) of the Ministry of the Environment, Japan partially supported this study.
References
Bianchi FJJA, Booij CJH, Tscharntke T (2006) Sustainable pest regulation in agricultural landscapes: a review on landscape composition, biodiversity and natural pest control. Proc R Soc B 273:1715–1727
Canhos VP, Souza S, Giovanni R, Canhos DAL (2004) Global biodiversity informatics: Setting the scene for a 'new world' of ecological forecasting. Biodivers Inform 1:1–13
Dubois O (2011) The state of the world's land and water resources for food and agriculture: Managing systems at risk. Earthscan by FAO, Rome
Frison E, Cherfas J, Hodgkin T (2011) Agricultural biodiversity Is essential for a sustainable improvement in food and nutrition security. Sustainability 3:238–253
Garibaldi LA, Steffan-Dewenter I, Winfree R, Aizen MF (2013) Wild pollinators enhance fruit set of crops regardless of honey bee abundance. Science 339: 1608–1611
Landis DA, Wratten SD, Gurr GM (2000) Habitat management to conserve natural enemies of arthropod pests in agriculture. Annu Rev of Entomology 45:175–201
Osawa T, Kurihara T, Nakatani Y, Yoshimatsu S (2011) Improvement of biodiversity information and utilization: a case study in developing an insectarium browsing system based on Internet technology. Japanese J of Conserv Ecol 16: 231–241 (in Japanese with English summary)
Osawa T, Yamanaka T, Nakatani Y (2013) Establishing a canonical procedure for collecting biodiversity information from citizen scientists using mobile phones. Japanese J of Conserv Ecol 18:157–165 (in Japanese with English summary)
Osawa T (2013) Monitoring records of plant species in the Hakone region of Fuji-Hakone-Izu National Park, Japan, 2001–2010 Ecol Res 28:541
Purcell AH, Almeida RP (2005) Insects as vectors of disease agents. In: Encyclopedia of Plant and Crop Science (Goodman RM, ed.) Taylor and Francis, London
Sasaki T, Katabuchi M, Kamiyama C, Shimazaki M, Nakashizuka T, & Hikosaka K (2014) Vulnerability of moorland plant communities to environmental change: consequences of realistic species loss on functional diversity. J of App Ecol 51:299–308
Tilman D, Knops J, Wedin D, Reich P, Ritchie M, & Siemann E (1997) The influence of functional diversity and composition on ecosystem processes. Science 277:1300–1302
Triplehorn CA, Johnson NF, Borror DJ (2005) Introduction to the study of insects (7th Edition) Thompson Brooks/Cole
Thrupp LA (2000) Linking agricultural biodiversity and food security: the valuable role of agrobiodiversity for sustainable agriculture. International Aff 76:283–297
Wieczorek J, Bloom D, Guralnick R, Blum S, Doring M, Giovanni R, Robertson T, Vieglais D (2012) Darwin Core: An evolving community-developed biodiversity data standard. PLoS ONE: e29715. doi: 10.1371/journal.pone.0029715
Yamamoto S, Kusumoto Y (2008) Strategy of biodiversity inventory for quantitative assessment of rural environmental change. J of Rural Plan Assoc 27:26–31. (in Japanese with English summary)
Vergara CH, Badano E I (2009) Pollinator diversity increases fruit production in Mexican coffee plantations: the importance of rustic management systems. Agric Ecosyst Environ, 129:117–123
Zak JC, Willig MR, Moorhead DL, & Wildman HG (1994) Functional diversity of microbial communities: a quantitative approach. Soil Biol and Biochem 26:1101–1108